|
Stable conjugate base, the fluoride anion is the That fluorine is the best at stabilizing a negativeĬharge and that makes this the most stable conjugate base. It likes to have electrons around it and therefore it makes sense Table you know you increase in electronegativity with fluorine being the most electronegativeĮlement and the most electronegative elementĪttracts electrons the most. That's the same trend asĮlectronegativity so if you look at carbon, nitrogen, oxygen,Īnd fluorine as you move to the right on the periodic With a negative charge and we had the most stable conjugate base. We go to oxygen withĪ negative one charge. Negative charge, right? This has a negative charge on carbon and this was our least stable conjugate base. Trend by looking at the element that has the Stability of the conjugate base and we can explain that The most stable conjugate base so as we move to the right we're increasing in the stability. Strongest acid must have the most stable conjugate base. Is the strongest acid out of these four and the Proton, then these electrons would be left behind on the fluorine to form the fluoride anion. The hydroxide anion as our conjugate base with a negative one formal charge on the oxygen. Proton these electrons are left on the oxygen to form So the nitrogen has a negative one formal charge. Proton, then these electrons are left on the nitrogen If we take this protonįrom methane, then these electrons are left behind on the carbons. So let me go down here and we'll get some more room. The conjugate bases for all four of these compounds 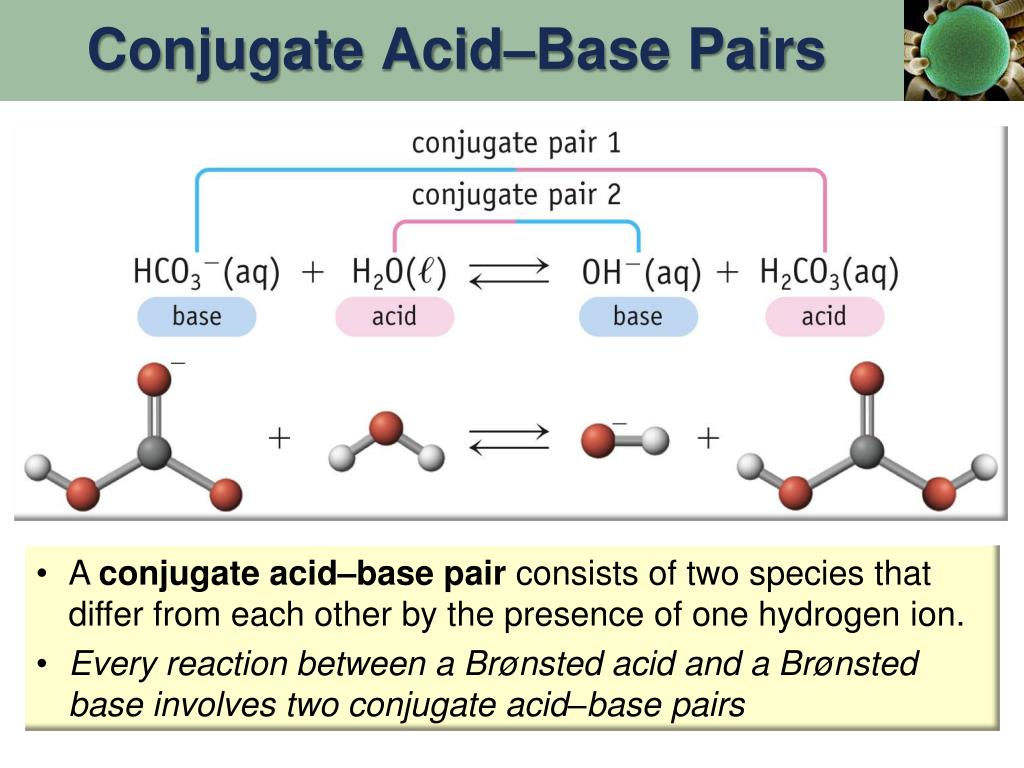
And if H-F is the strongestĪcid out of these four, then H-F must have the We see an increase in acid strength so H-F is the strongest acid out of these four. Therefore, as we go to the right we see an increase in acid strength. Right we see a decrease in PKA values from 48 to 36 to 16 to three. Value, the stronger the acid so as we move to the And if we look at water, the PKA for this proton is about 16 and finally for H-F, this proton has a PKA of about three. Let's use that concept and let's look at these four compounds down here. Stable the conjugate base, the stronger the acid. The more stable the conjugateīase, the more likely the acid is to donate a proton. 
Look at the stability of the conjugate base. To determine the acidity of a compound, you can If A minus is stable, then H-A is more likely to donate this proton. This bond, so the electrons in magenta are left behind Generic acid and donates this proton, the electrons in
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
